Industry seeks more economical fuel cells
Fuel cells, like other alternative forms of energy, offer a number of positive features compared to fossil fuels-based power generation.
Typically powered with hydrogen, they don’t release the climate-changing carbon associated with combustion. They are quiet, durable and—produced wisely—don’t create air or water pollution. And for a nation concerned about energy independence, they help wean us from our dependence on foreign oil.
Markets for fuel cells are growing. Larger stationary fuel cells provide power for buildings and cell towers. Smaller fuel cells do the same for electric vehicles and forklifts.
But fuel cells also have a down side. For one, fuel cell power is more expensive than the gasoline power that keeps cars and trucks on the road, primarily due to the costly catalysts needed to facilitate the chemical reactions taking place. (For more information on catalysts, see sidebar below.)
For another, the hydrogen we now generate comes mostly from feedstocks of fossil fuels, principally natural gas. Not only is this process expensive; it continues to exacerbate climate change. In other words, while the fuel cells themselves don’t introduce greenhouse gases into the environment, the production of hydrogen from natural gas does. If we are to make the hydrogen fuel and fuel cells at a lower cost and with a low carbon footprint, we must find alternative feedstocks and less expensive catalysts.
A computational effort led by Amra Peles of United Technologies Research Center (UTRC) is pushing the limits of lower-cost, genuinely renewable hydrogen production and use for fuel cells. United Technologies is among the country’s 20 largest manufacturers, with nearly $60 billion in sales in 2011. The company provides a broad range of high-technology products and services to the global aerospace and building systems industries. It is well known for its aerospace technology brands Pratt & Whitney (aircraft engines) and Sikorsky (helicopters). It is also a leader in the design and manufacture of innovative systems for heating and air conditioning (Carrier brand), lighting, and energy management of buildings, all ripe areas for fuel cell application. UTRC pursues technologies for improving the performance, energy efficiency and cost of UTC products and processes.
Working with the Cray XT5 Jaguar supercomputer at Oak Ridge National Laboratory (ORNL), Peles and colleagues explored both the use of biomass as a low-carbon, renewable hydrogen feedstock, and the effectiveness of lower-cost nickel as a replacement catalyst for platinum. Success in this arena will provide a renewable, cost-effective, nearly carbon-neutral way to produce hydrogen and contribute to the development of improved, long-lived, cost-effective fuel cells.
Their work has been published in Nano Letters, the Journal of Physical Chemistry Letters, and the Journal of Materials Science.
The Problem with Platinum
Platinum is the most effective catalyst yet found for producing hydrogen from biomass. But its expense is a key reason the cost of fuel cells remains high. Peles and her colleagues are investigating alternative, less expensive replacements for platinum, including the use of nickel to produce hydrogen from biomass. They are also delving into the opportunities offered by changes in the catalytic performance of a material when its size changes from bulk to nanometer dimensions.
Materials at the nanoscale display new phenomena that play a negligible role at larger scales. Because the particles are so small, their behavior is governed largely by the rules of quantum mechanics in a phenomenon known as quantum size effect. In addition, the role of surfaces and interfaces becomes predominant.
Peles needed to understand key nanoscale properties for both platinum and nickel. Computer simulation on very large-scale systems is the most effective way to examine chemical properties at the nanoscale level, given the number of atoms that must be represented in the models and the complexity of solving the quantum-mechanical theories describing chemical reactions on catalytic materials. The computational requirements greatly exceeded UTRC’s internal computing resources.
It Takes a Big Computer to Study Small Particles
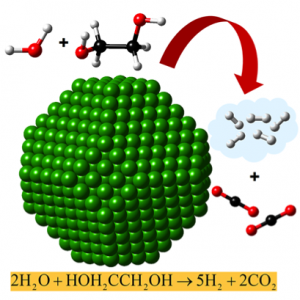
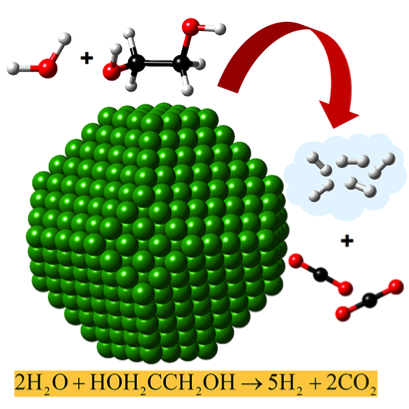
This image shows the reforming of ethylene glycol to produce hydrogen. In the presence of the catalyst (green), water and ethylene glycol react to form hydrogen and carbon dioxide. The balls represent atoms; black is carbon, red is oxygen, and white is hydrogen. Image courtesy United Technologies Research Center.
Peles turned to ORNL’s Jaguar, one of the Department of Energy’s leadership high-performance computing systems and one of the most powerful in the world. Through ORNL’s HPC Industrial Partnerships Program, Peles tapped Jaguar’s power to dramatically expand UTRC’s understanding of catalytic processes at the nanoscale level. Peles was joined in her effort by colleagues Minhua Shao, Sean Emerson, Rhonda Willigan and Tom Vanderspurt.
Peles and her team used the ethylene glycol molecule as a proxy for biomass in their simulations of biomass reforming because it exhibits the full range of chemical bonds found in biomass. They uncovered opposing trends when examining the catalytic reactions of platinum and nickel with this molecule. Platinum prefers a direct reforming pathway that converts the molecule to hydrogen, while nickel prefers a pathway that leads to the formation of methane—a very serious greenhouse gas—in addition to hydrogen.
When examining hydrogen conversion reaction in fuel cells, they also had to balance two competing challenges. On the one hand, the catalyst’s particles should be as small as possible in order to maximize the amount of exposed surface area. On the other hand, particles below a certain size stop catalyzing the reaction and, therefore, become useless.
“Understanding how the particle size of a nanostructured catalyst affects activity is a longstanding problem,” Peles explained. “Because platinum is so expensive, you want to get the best possible ratio of reaction sites on the surface, maximizing surface area. We want smaller particles, but there’s a limit to how small you can get. By studying particles in different sizes we are able to provide an understanding of the limits of precious metal utilization.”
Peles and her colleagues explored these reactions on Jaguar with two specific codes. LAMMPS, for Large-scale Atomic/Molecular Massively Parallel Simulator, is a molecular dynamics application simulating the interaction of atoms and molecules. VASP, for Vienna Ab initio Simulation Package, uses a quantum mechanical approach known as density functional theory to provide an atom-by-atom understanding of different materials and combinations of materials used as catalysts.
The version of VASP Peles used was optimized for Jaguar by ORNL’s Paul Kent. Peles said she was interested in working on Jaguar at least in part because it would give her access to Kent’s optimized version of the code, which she said was the most robust and high-performing iteration available for her research.
“Our aim was to exploit the scalability of VASP and push the limits of the model sizes we can simulate,” Peles explained. “VASP on Jaguar was probably the best optimized compilation of this program that exists. Using VASP on Jaguar we were able to increase our model size by a factor of 10.”
Success with Some Compromise
Access to VASP and Jaguar paid off. Using first-principles atomistic modeling, the team gained insight into the factors that control the activity and selectivity of hydrogen using platinum and nickel catalysts, as well as the optimum size for the catalytic particles in fuel cells. So what’s the best size? Peles and her colleagues determined that 2 to 3 nanometers will provide the most exposed surface while ensuring that particles are effective.
And how did nickel fare in producing hydrogen compared to pricey platinum? Peles and her team discovered that nickel could be an adequate substitute for platinum … with an important caveat: that disturbing tendency to produce methane along with hydrogen in the catalytic process.
“Platinum is a very good catalyst,” Peles noted, “and prefers the formation of hydrogen and CO2, which are two desirable major products. Nickel, on the other hand, has a tendency to produce a lot of methane. Our conclusion is that these two metals have different pathways toward the production of hydrogen.”
Further collaboration with a team of UTRC experimentalists revealed that nickel becomes a better catalyst for this process when it is working in a high-pH environment. “We determined that when the environment is more alkaline the production of hydrogen is boosted and methane lowered,” Peles said. So while nickel still doesn’t match platinum in its effectiveness in hydrogen production—“there is some compromise,” states Peles—it becomes an adequate, and economically viable, substitute in an alkaline environment. In fact, she said, the methane produced along the way can be used in the process to heat the chemical reactor in which the catalysis takes place.
New Understanding … New Opportunities
Access to Jaguar and a scalable compilation of the VASP code available only at the Oak Ridge Leadership Computing Facility allowed Peles and her teammates to gain new insights into how platinum and nickel catalysts produce hydrogen from biomass, along with other byproducts like methane.
“Our ability to study larger, more realistic and complicated models with Jaguar allowed us to uncover property changes and features of platinum- and nickel-based catalysts that were unknown to us previously and accessible only at the nanoscale.” explained Peles.
They further discovered with their experimentalist colleagues that nickel’s ability to produce hydrogen was significantly increased in an alkaline environment, a critical insight when considering whether a less expensive, nickel-based catalyst is a feasible alternative for the platinum catalysts used for hydrogen production from biomass. Peles and her team also confirmed the optimum size for the catalytic particles in fuel cells to ensure their effectiveness.
All of these discoveries will contribute significantly to UTRC’s and the nation’s efforts to produce more commercially viable fuel cells from renewable resources so we can reduce our dependence on imported fossil fuels.
Related publications
Shao, M., et al. 2011. “Electrocatalysis on Platinum Nanoparticles: Particle Size Effect on Oxygen Reduction Reaction Activity,” Nano Letters, 11, pp 3714–3719.
Shao, M., et al. 2010. “Enhanced Oxygen Reduction Activity of Platinum Monolayer on Gold Nanoparticles,” Journal of Physical Chemistry Letters, 2, pp 67–72.
Peles, A., 2012. “GGA+U method from first principles: application to reduction–oxidation properties in ceria-based oxides,” Journal of Materials Science, 47, pp 7542–7548.