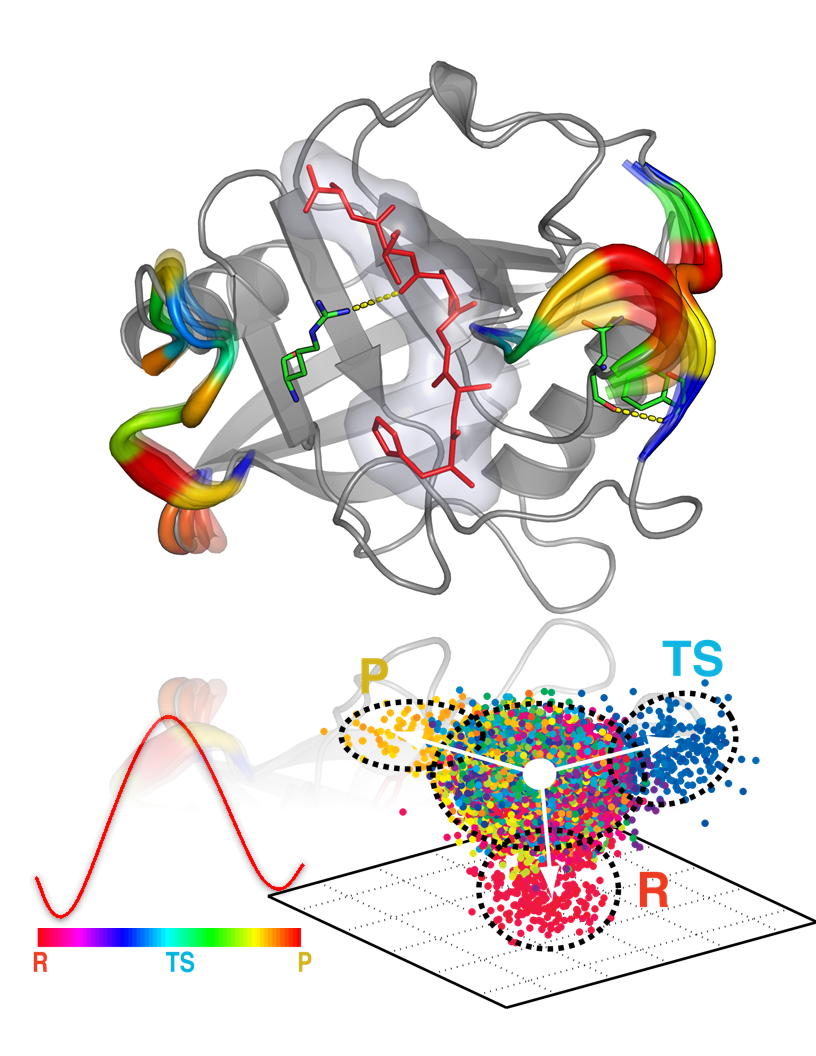
Identification of reaction promoting dynamics and conformational sub-states.
Titan enables researchers to uncover the mechanisms behind nature’s greatest catalysts
Few things are more essential to living organisms than enzymes, those plentiful molecules that make possible the biological reactions necessary for life, such as generating energy from food or more complex tasks like the synthesis of DNA. The vast majority of enzymes belong to a class of biomolecules known as proteins.
Enzymes are of particular importance across multiple scientific arenas for a number of reasons, not the least of which is their distinction as nature’s greatest catalysts. Enzymes excel at accelerating countless biochemical reactions important to a wide spectrum of science, including applications in medicine, clean energy, and industrial manufacturing. Exploiting their high catalytic efficiency could be a game changer for any and all of these fields.
Enter Titan, Oak Ridge Leadership Computing Facility’s flagship supercomputer, which is currently ranked as America’s fastest. The hybrid-architecture Cray XK7 is the weapon of choice for a team of researchers led by Oak Ridge National Laboratory’s Pratul Agarwal seeking to obtain key insights into the fundamental underpinnings of how enzymes work and, in particular, what makes them such great catalysts.
“Using Titan has enabled us to create high-quality predictive models of proteins and enzymes,” said Agarwal.
His team is modeling more than a dozen enzymes in an attempt to figure out what behaviors they have in common, especially those related to their catalytic capabilities.
“Titan allows us to sample timescales relevant to the catalytic function of enzymes spanning 12 orders of magnitude, from femto- to millisecond, using a few computational tricks,” said Agarwal. “It also allows us to look at similar enzymes from single-cell bacteria in complex life forms such as humans.
A tall order for sure, but the team’s work is beginning to pay off. Thanks to information gleaned from Titan, Agarwal and his collaborators have developed a hyper-catalytic version of the industrial enzyme lipase B, which showed a 3,000-percent increase in catalytic efficiency in one round of enzyme engineering and testing. Other state-of-the-art approaches such as directed evolution may take five or six rounds of iterative engineering to achieve this kind of increase.
Changing shape
Enzymes are as not rigid as the structures depicted in biochemistry textbooks. They are constantly shifting and changing shape in fractions of a second. Certain shapes promote catalysis more than others, and it is these that Agarwal’s team hopes to pin down. If the catalytic suitability of each conformation (or shape) for catalytic function can be calculated, then novel enzymes can be engineered to hasten chemical reactions critical to countless areas of science, enabling breakthroughs at breakneck speed.
Using two molecular dynamics applications—the Large-scale Atomic/Molecular Massively Parallel Simulator (LAMMPS) and Assisted Model Building with Energy Refinement (AMBER)—the team is able to see how enzymes move over time in relation to function.
The team is modeling a large set of enzymes and attempting to figure out which advantageous behaviors are common to all of them in an effort to build a biophysical model of enzyme catalysis.” The hope is that these traits can then be put to use in any number of other systems.
Ultimately three facets of enzymes are critical to understanding their catalytic efficiency: (1) how an enzyme’s conformation changes (or the small shifts in structure), (2) how changes in conformations play a role in the catalytic mechanism (in particular, the search is on for the transient substates that occur for a thousandth or even a millionth of a second but are vital for the enzyme’s function), and (3) how nature has conserved these critical dynamic features of the enzyme over evolution between bacteria to humans.
These changes in an enzyme’s conformations are rapid and therefore difficult—and expensive—to capture experimentally, and even the most expensive experimental techniques can provide information over only small range of timescales. With simulation, however, and with systems on the scale of Titan in particular, these conformational fluctuations can be obtained over the femto- to millisecond timespan. Once gathered, this information can then be compared to that obtained from experimental sources.
This comparison, with data collected from nuclear magnetic resonance and neutron scattering techniques, is beginning to provide the team with vital clues regarding what traits make certain enzymes better catalysts than others, and what parts of the enzymes drive catalysis. “The findings indicate that in addition to the well-known structural features, the dynamic features that support the function are also present across a number of enzyme species,” said Agarwal
GPUs make the difference
“I never thought it would be possible to investigate enzymes over such a wide range of timescales,” said Agarwal. This capability is enabled by Titan’s GPUs, which work in concert with the more traditional CPUs to crunch numbers, or in this case partial differential equations, like few systems can.
In fact, Agarwal’s team was among the first to use Titan’s hybrid architecture, and Agarwal himself worked closely with GPU manufacturer NVIDIA to optimize the molecular dynamics codes to the GPUs, an experience that allowed the team to begin production runs almost immediately, he said.
These production runs occupy anywhere from 5,000 to 45,000 of Titan’s nearly 300,000 compute cores. The “ensemble” simulations allow simultaneous sampling of the various enzymes’ conformational states, Agarwal explained, meaning that his team can arrive at the optimal catalytically relevant conformations more rapidly than ever before. Moreover, multiple simulations allow for the collection of statistically meaningful information, something far out of reach for single, one-time observations.
And when it comes to something as complex as investigating the evolution of enzyme molecules, time is of the essence. Because there are literally thousands of enzymes, calculating various substates for even a small set of these enzymes and their effects on one another is clearly a daunting task. Thanks to Titan, however, it’s not as daunting as it could be.
“The quantitative estimations for this research require extensive sampling, which is enabled by Titan,” said Agarwal.
Related Publications:
Protein Conformational Populations and Functionally Relevant Substates. Accounts of Chemical Research, Vol. 47, No. 1, 2014,149–156.