by Argonne National Laboratory
Key Takeaways
- Carbon is one of nature’s most versatile elements, capable of forming materials that are stronger than steel, lighter than plastic and adaptable to countless applications.
- Using powerful supercomputers and artificial intelligence and drawing on expertise in multiple disciplines, Argonne scientists are learning to predict and control how tiny carbon crystals respond under extreme conditions.
- Their work, published in the journal Carbon, could lead to breakthroughs in medicine, energy storage and national security while showcasing the power of supercomputing and artificial intelligence in modern science.
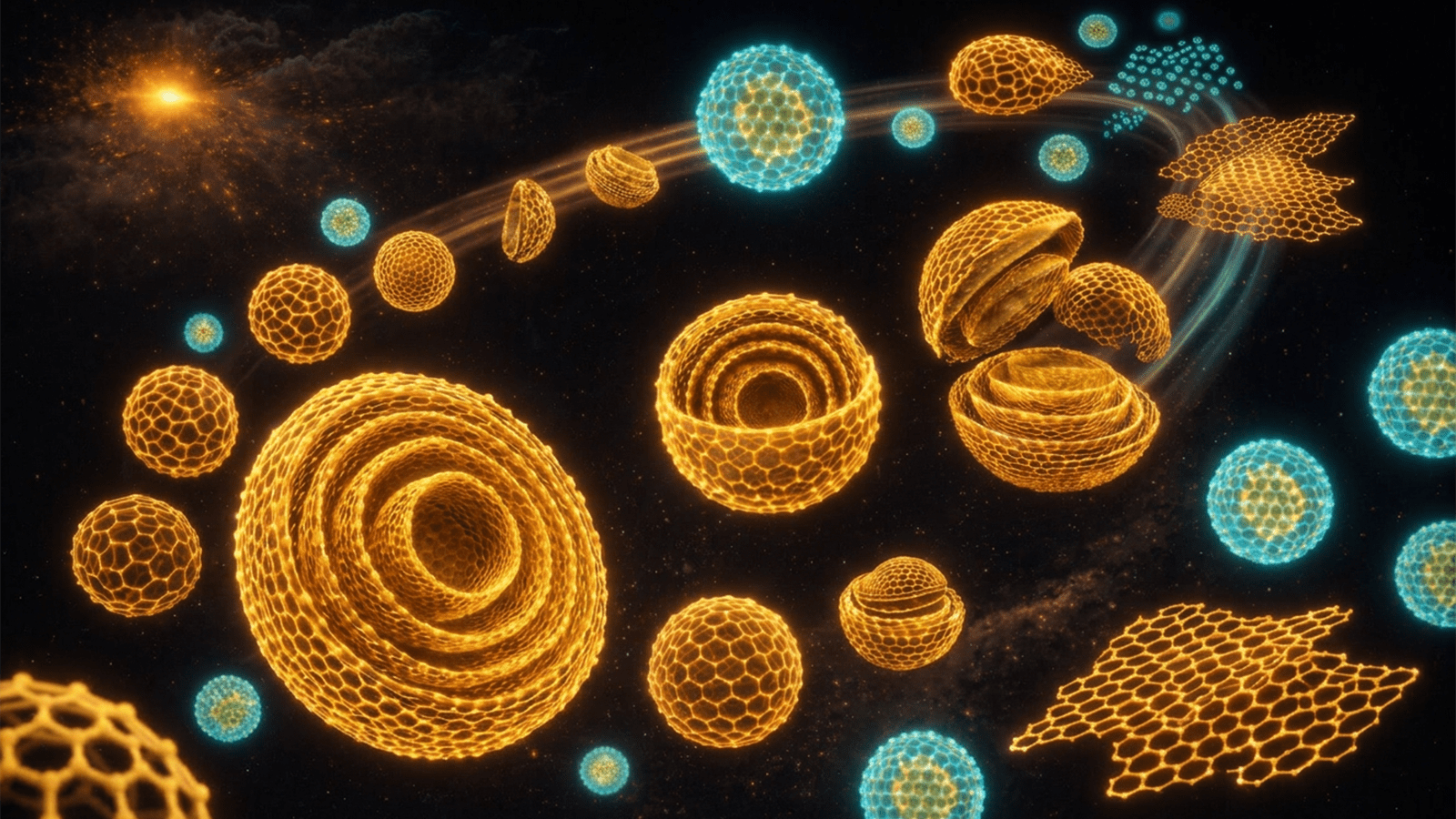
Carbon is everywhere. It forms the graphite in pencils, the diamonds in jewelry and the molecules that make up every living thing. But under extreme conditions — like the heat and pressure of intense explosions — carbon can transform into exotic nanometer-sized structures called nanocarbons. These materials are often stronger than steel, lighter than plastic and adaptable for uses in medicine, energy and national security.
Scientists at the U.S. Department of Energy’s (DOE) Argonne National Laboratory and partner universities are uncovering how carbon behaves in extreme environments. By combining physics, chemistry, supercomputers and artificial intelligence (AI), researchers are learning to predict and control the ways that carbon atoms rearrange themselves under intense heat and pressure. Their discoveries open the door to designing materials before they are ever made in a laboratory.
Their findings are now out in the journal Carbon.
“Carbon is remarkably versatile, and under extreme conditions, it reveals entirely new structures,” said Eliu Huerta, lead for translational AI in Argonne’s Data Science and Learning division. “By integrating physics-based modeling, AI and exascale computing, we can predict how carbon assembles at the nanoscale and use that insight to design advanced carbon materials with properties tailored for real-world applications.”
The research team focused on nanodiamonds, tiny diamond crystals only a few millionths of a millimeter across. Thousands of these crystals could fit inside the width of a human hair. The particles form in explosive environments, where temperatures soar higher than the surface of the sun and pressures rise to millions of times that of Earth’s atmosphere.
What happens next — how the material cools and how the pressure drops — determines whether the nanodiamond stays a diamond or transforms into other carbon forms, such as layered sheets or hollow structures.
Instead of relying solely on conducting costly and dangerous experiments to understand nanodiamond behavior, the team used Argonne’s Aurora supercomputer to simulate these transformations atom by atom. Aurora is the flagship computer of the Argonne Leadership Computing Facility, a DOE Office of Science user facility. They also used the Frontier supercomputer at DOE’s Oak Ridge National Laboratory. The Delta and DeltaAI systems at the National Center for Supercomputing Applications at the University of Illinois Urbana-Champaign were also used to perform some of the simulations of the carbon structures.
The computer models act like ultrahigh-speed cameras, letting scientists watch carbon atoms move and bond in slow motion as conditions change.
The researchers’ simulations revealed that the “recipe” of cooling and pressure release strongly influences the final structure of the material. Rapid cooling tends to preserve the diamond form, while slower cooling allows carbon atoms to rearrange into layered shells and curved structures.
To turn these discoveries into a practical design tool, the scientists trained AI models on the massive simulation data they produced. The models learned to recognize patterns between temperature, pressure and the final shape of the carbon material. With this approach, the computer could predict what kind of nanocarbon will form from a given set of conditions. This means scientists can design new materials on a computer first, rather than relying on lengthy trial-and-error experiments in the lab, saving both time and research funds.
“Understanding how carbon atoms form various nanoparticles has long been a fundamental scientific question for me, and Aurora enables us to explore along this path atom by atom and condense that knowledge into mathematical models,” said Xiaoli Yan, a postdoctoral researcher in Argonne’s Data Science and Learning division.
Different carbon structures have different uses. Nanodiamonds can be used in quantum sensors and medical imaging. Onion-like carbon particles made of layered shells are promising for storing electrical energy. Other tiny carbon forms can glow under light and may be useful in light-sensitive devices or biological imaging.
Or consider hollow carbon shells. Compared to solid particles or flat flakes of carbon, hollow shells are better at carrying microscopic cargo and slipping inside cells. The shape makes them perfect for one day carrying medicines directly into diseased cells.
By understanding how to guide carbon into specific shapes, scientists can tailor these materials for specific applications.
“What’s exciting is that supercomputers let us simulate extreme conditions, and AI turns that knowledge into the ability to design extraordinary, tailorable materials,” said Millie Firestone, a senior scientist in Argonne’s Physics division.
This research also has important implications for national security. Carbon plays a key role in many defense and industrial technologies, especially those involving high-energy environments. Knowing how carbon behaves under extreme conditions helps improve models of explosives and protective materials. It also supports the design of stronger coatings, lighter armor and more resilient components for use in harsh environments, from deep underground to outer space.
“At its heart, this work blends several fields into one powerful approach. Physics explains how atoms move. Chemistry explains how bonds form and break. Computer science provides the tools to simulate millions of atoms at once. AI ties everything together by learning from the data and making predictions,” Huerta said.
While computer-aided materials design has existed for years, the integration of AI now allows computers to actively learn, adapt and help drive discovery.